Elemental analysis is an important technique for determining the composition and purity of compounds, but a new study has raised concerns that the data is prone to manipulation.
Christian Kowol and colleagues from the University of Vienna, Austria, analysed six commercially available high purity compounds, using two different elemental analysis instruments and three additional independent elemental analysis services. They found typical deviations from theoretical values were 0.05–0.20%. They also surveyed the literature and found that ~5–10% of publications reported deviations ≤0.10% from theoretical values for most or all data, despite newly developed compounds not necessarily having fully optimised synthesis and purification processes. Considering these deviation surveys together, the researchers say, indicates a significant number of studies contain unlikely elemental analysis values.
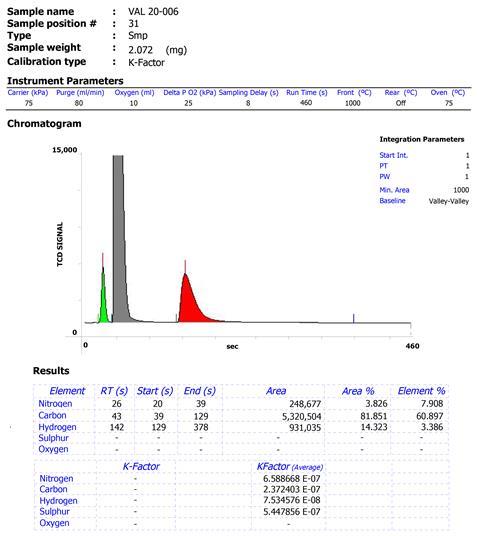
‘The current practice of just reporting elemental analysis numbers without providing any raw data or reports leaves room for suspicious reporting and is in stark contrast to the standard practices for reporting other characterisation data,’ comments Christine Thomas, an inorganic chemist from the Ohio State University, US, who was not involved in the study.
‘Whenever we talked to older professors, they told us, ”yes, we know this already from 20 years ago, there was exactly the same situation.” … When you do a lot of work to get pure compounds to have good science and good data, it’s of course not really fair if others don’t do that,’ says Kowol.
Kowol’s team therefore has a proposal: researchers reporting elemental analysis data should include a figure of the original data with peak integrations, alongside details of the standards used for calibration, instrumentation, number of measurements and other key experimental information. Kowol says presenting the data is quite straightforward and that if journal and reviewer expectations change, collating this additional information should become even easier. ‘It was definitely the aim of the work to change something here in the future.’ He suggests that reviewers refer to his team’s study when requesting such information from paper authors.
‘Many researchers do not have access to an analyser in their own labs or institutions and rely on shipping samples to external service facilities that do not typically provide that type of detailed report and experimental information. Making such detailed information a requirement for publication would, therefore, be a burden and perhaps an insurmountable obstacle to many researchers,’ cautions Thomas. ‘If analysis was performed externally, I think it would be reasonable to ask the authors to include the company’s report as supporting information.’ Kowol says that if journal and reviewer expectations change, collating this additional information should become easier. ‘When companies who sell such instruments see that it’s more common that you eg make such a graph and put it somewhere, they will adapt it so it’s very easy to do this and to have all the information already on one page and you just have to copy and paste it.’
William Tolman, of Washington University in St Louis, US, and editor-in-chief of the journal Inorganic Chemistry, says the recommendations for reporting requirements are intriguing. ‘Our team of editors regularly discusses and updates the journal’s guidelines to ensure data quality and to minimise the burden on authors and reviewers, and we will take a closer look at reporting elemental analyses.’