### Introduction to CRISPR
CRISPR is an innovative mechanism that is changing lives and creating significant impact in the healthcare sector. CRISPR, which represents “Clustered Regularly Interspaced Short Palindromic Repeats,” is present in prokaryotes—small, unicellular entities devoid of organelles. These sequences exist within the genomes of prokaryotes, which compartmentalize a cell’s DNA in clusters.
As a genetic modification tool, CRISPR is utilized to alter certain segments of DNA to address serious illnesses. It is particularly effective as it assists the body in combating viral attacks by integrating the invading DNA into its own genetic makeup, enabling it to identify and eliminate the virus in subsequent encounters. Notably, in comparison to other genetic modification techniques, CRISPR is significantly more accurate and simpler to program, facilitating an uncomplicated sequence alteration process. While traditional gene-editing instruments generally depend on a single protein, CRISPR employs RNA-guided targeting in conjunction with the Cas9 enzyme.
—
### How CRISPR Works
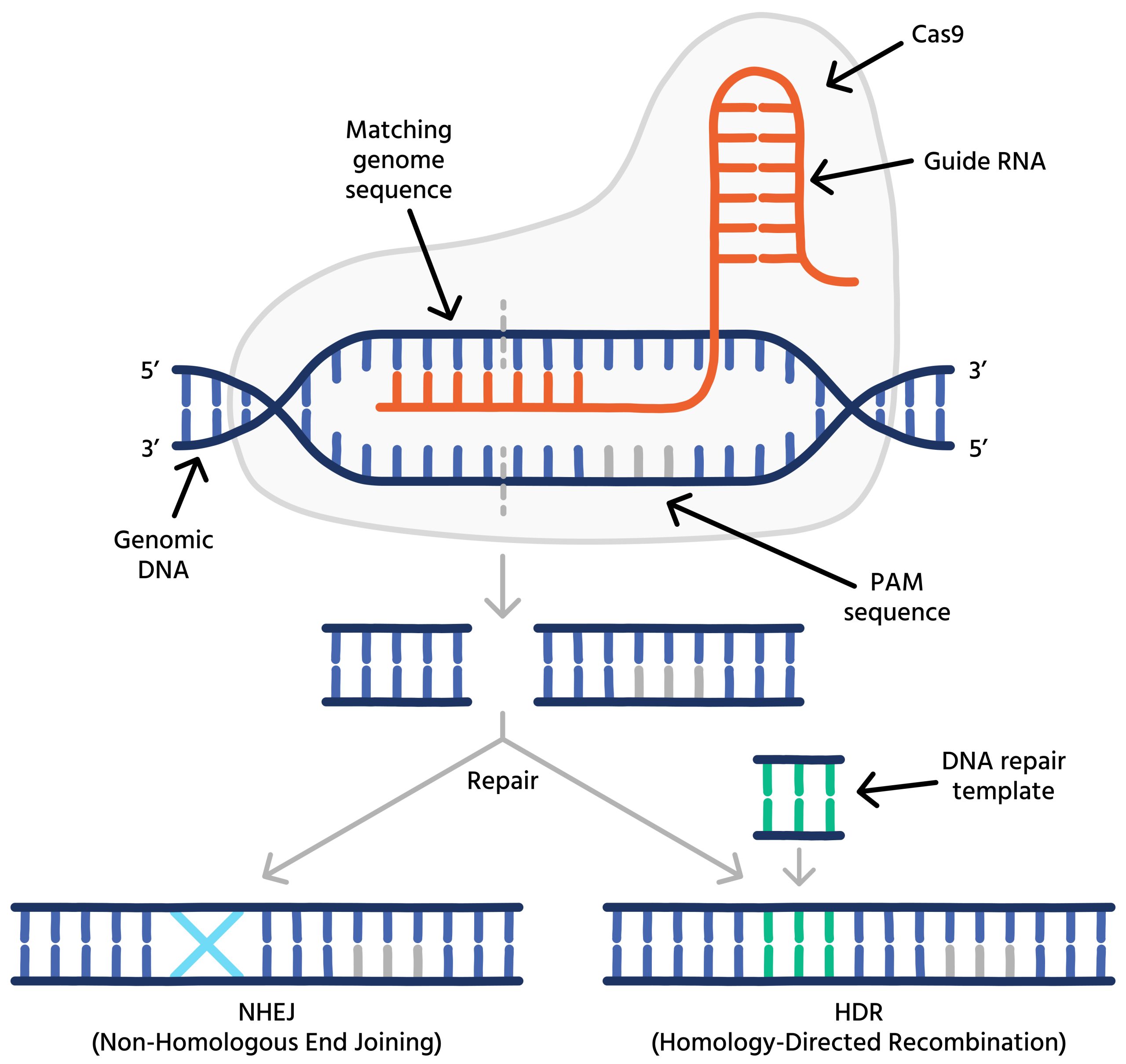
CRISPR operates by utilizing a native defense system discovered in bacteria that equips them to detect and eliminate viral threats. When a virus infiltrates a bacterial cell (a prokaryote), the bacterium retains a fragment of the invader’s DNA within its own genome as a genetic “memory.” This allows the bacterium to recognize and react more efficiently to future outbreaks.
In the field of genetic modification, this mechanism is fine-tuned using two essential components: the Cas9 enzyme, which acts as molecular scissors to cleave DNA, and guide RNA, which steers Cas9 to the particular genetic sequence requiring modification. Once the designated DNA is severed, the cell’s inherent repair processes engage, permitting scientists to implement alterations to the genetic makeup.
In contrast to earlier technologies that relied on difficult-to-reprogram proteins, CRISPR’s RNA-driven approach is more adaptable, straightforward to design, and highly precise. This ease and accuracy have enabled CRISPR to find applications in medicine, agriculture, production, and microbiology—such as modifying microbes to enhance output. However, as this article examines, with increasing capabilities arise escalating ethical dilemmas, especially concerning germline editing and genetic enhancements.
—
### World’s First Patient of a Personalized CRISPR Treatment
In February 2025, the first-ever personalized CRISPR treatment was administered to a baby named KJ to address a deficiency in Carbamoyl Phosphate Synthetase 1 (CPS1)—an enzyme crucial for converting ammonia (generated during protein degradation) into urea. A team led by Dr. Rebecca Ahrens-Nicklas and Dr. Kiran Musunuru at the Children’s Hospital of Philadelphia formulated this treatment following extensive research in genetic editing and collaboration with other healthcare professionals.
Their focus was on conditions affecting the urea cycle, which results in the accumulation of toxic ammonia, hence harming organs including the brain and liver. They customized the treatment specifically for KJ’s variant of CPS1 deficiency using preclinical data on similar cases.
Up to this point, the only CRISPR therapies endorsed by the U.S. FDA have been for more prevalent conditions like sickle cell disease and beta thalassemia, impacting tens or hundreds of thousands of individuals. In KJ’s situation, his treatment was crafted within half a year of his birth, targeting his unique CPS1 variant. The team developed a base editing therapy administered through lipid nanoparticles to his liver to rectify the faulty enzyme.
The February intervention was the inaugural of three doses; KJ received the subsequent two in March and April 2025. As of his last administration, he has shown no significant adverse effects, displays improved tolerance to dietary protein, and necessitates less medication to regulate ammonia levels. Although ongoing observation will be required, Ahrens-Nicklas asserts that the early results are encouraging.
—
### Ethical Considerations of CRISPR
Like any pioneering technology, CRISPR presents intricate ethical considerations. While its primary aim is to modify somatic cells to treat ailments, it can also be applied to gametes, venturing into the contentious area of germline editing. Modifying DNA that will be passed down to future generations is frequently viewed as unethical—especially when conducted to enhance traits rather than to remedy diseases.
In light of these apprehensions, researchers have momentarily barred germline editing until its ethical and social ramifications are more clearly understood. The unpredictability of genetic alterations, along with the permanence of such changes across generations, raises vital inquiries about where to set boundaries. What started as a groundbreaking concept has evolved into a formidable reality, stirring discussions on who gets to determine its parameters.
CRISPR’s steep costs and sophisticated infrastructure needs render it more attainable in affluent, developed nations—particularly in the West. As treatments stay costly, accessibility becomes confined to the wealthy, intensifying socioeconomic disparities. The potential for utilizing CRISPR to create so-called “