Manipulating coulombic forces could provide a different method to influence selectivity in electrocatalytic processes. Rather than attempting to promote the desired reaction, this method – referred to as electrostatic decatalysis – functions by inhibiting competing pathways through the electrostatic engineering of the electrode surface, a concept that has been mostly neglected in the pursuit of solutions for energy and environmental issues.
In various electrocatalytic reactions, undesirable side reactions compete with the intended transformation, consuming both reagents and energy, decreasing efficiency, and ultimately diminishing selectivity. Traditional attempts to enhance these reactions often center on redesigning the catalyst’s structure. However, such changes frequently involve sacrifices, improving the catalyst’s activity for both the desired and competing reactions, without controlling selectivity.
Electrostatic decatalysis adopts another approach. While coulombic (electrostatic) forces are well known in electrochemistry, they have not been previously utilized to intentionally suppress unwanted reactions. This idea, formulated by Musthafa Ottakam Thotiyl at the Indian Institute of Science Education and Research, aims to manipulate long-range coulombic interactions at the electrode surface to obstruct undesired pathways. Ottakam Thotiyl compares this to a gate system: the gate is open for only the desired reaction and closed for the competing reaction.
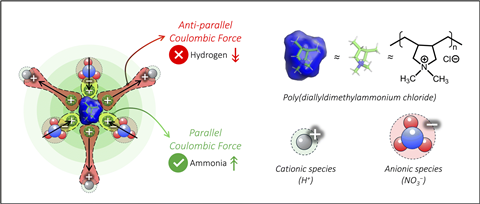
The research team experimented with this concept on the electrochemical nitrate reduction reaction, where the hydrogen evolution reaction typically competes. They treated carbon nanotube electrodes with either the cationic ionomer poly(diallyldimethylammonium chloride) (PDDA), imparting a positive charge to the surface, or the anionic ionomer Nafion, resulting in a negative charge. The positively charged PDDA suppressed the hydrogen evolution reaction and enhanced ammonia production, while negatively charged Nafion promoted the hydrogen evolution reaction and inhibited ammonia production.
Utilizing various experimental techniques and, in partnership with a colleague at the University of Texas at Arlington, US, computational modeling, the researchers investigated the reaction kinetics thoroughly. By methodically eliminating influencing factors such as wettability, electronic conductivity, and surface area, they verified that the enhanced selectivity stemmed from electrostatic interactions. The positively charged PDDA surface repels protons, restricting hydrogen evolution, while attracting nitrate ions to the electrode. The findings indicate that altering surface charge can direct competing reactions in opposite directions.
The team also showcased the method in a two-electrode ammonia synthesis system designed to replace the slow counter electrode oxygen evolution reaction with a ferrocyanide oxidation reaction. In this configuration, PDDA-modified electrodes accomplished energy savings exceeding 50% compared to a traditional nitrate reduction–oxygen evolution cell, attributed to advantageous electrostatic interactions.
‘The pH‑insensitive nature of this method makes it widely applicable under various conditions, unlike many current strategies constrained to specific pH ranges,’ remarks Heng Rao, an experimental electrochemist at Jilin University in China, who focuses on interfacial phenomena.
The team is now extending electrostatic decatalysis to additional reactions, including carbon dioxide conversion. However, Ottakam Thotiyl emphasizes that this strategy relies on charge: ‘This method is effective whenever the target substrate holds a charge. If the target substrate is uncharged, it will not function. It is based on coulombic repulsion, so the electrode must have a charge, and for repulsion to occur, the target should also possess a charge. That’s crucial.’