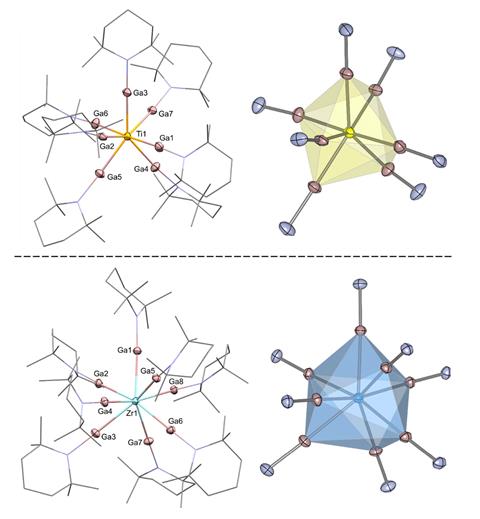
Chemists have recently succeeded in isolating the inaugural homoleptic titanium(0) and zirconium(0) complexes that possess a seven-coordinate structure, entirely stabilized by monodentate gallium ligands. These complexes stand out due to their exceptional stability, which is attained through cooperative gallium–gallium interactions, presenting an exciting new strategy for highly-reduced early transition metal systems.
Early transition metals such as titanium and zirconium are recognized for their high electropositivity and oxophilicity, usually remaining stable in elevated oxidation states. This poses a challenge for the formation of zero-valent forms, which often yield temporary, highly reactive entities that necessitate substantial stabilization.
The stabilization found in these innovative complexes arises from encircling the metal centers with seven monovalent GaTMP ligands (TMP = 2,2,6,6-tetramethylpiperidinyl). The gallium atoms leverage their expansive 4p orbitals to facilitate strong π-backbonding from the electron-rich metal centers, redistributing electron density more efficiently than traditional ligands. Simultaneously, the gallium centers partake in gallium–gallium covalent interactions, creating a stabilizing network of contacts throughout the ligand sphere. This cooperative bonding alleviates steric repulsion between the sizable ligands, delivering the essential stabilization for these atypical complexes.
The structural characteristics of these complexes were elucidated through a variety of methods, including single-crystal x-ray diffraction, NMR, IR/Raman, UV–vis, and high-resolution mass spectroscopies. Density functional theory calculations further clarified the gallium–gallium interactions and electronic attributes vital to their stability.
This development not only expands the chemistry surrounding early transition metals but also highlights the promise of metalloligands as cooperative frameworks. The researchers propose that this approach could be modified to stabilize other elusive low-valent species across the periodic table, potentially leading to highly reduced f-block complexes.