Two novel skeletal editing methods provide a means to generate a variety of nitrogen-containing rings. Nitrogen heterocycles are essential components in numerous pharmaceutical agents, with the researchers involved in these investigations stating that these synthetic approaches could facilitate the rapid discovery of prospective drug candidates.
Over 80% of new small molecule medications authorized by the US Food and Drug Administration from 2013 to 2023 included nitrogen heterocycles, including piperidines. Chemists frequently construct such rings via the Beckmann rearrangement, which entails reacting a cyclic oxime with an acid to yield an amide. Nonetheless, this method is limited by poor regioselectivity, necessitates harsh reaction conditions to convert the amide to the amine, and results only in ring expansion.
In addition, ‘indoles are crucial structural motifs in pharmaceuticals and natural products’, remarks Huiying Zeng from Lanzhou University in China. Modifying the pyrrole ring of an indole can influence the biological and physical characteristics of such compounds, but the ‘aromatic stability [of indoles] makes precise alteration of the core skeleton difficult’, according to Zeng.
Zeng’s team has now created a technique that intramolecularly transfers substituents to the C2 position of indole rings.1 Similarly, another team led by Lumin Zhang at the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences is employing an iodine-based catalyst to generate nitrogen-containing rings of various sizes and types from primary amines.2
‘These two [studies] exemplify the ingenuity and innovation that the idea of skeletal editing inspires,’ comments Richmond Sarpong at the University of California, Berkeley in the US, who did not participate in either study.
Versatile amines
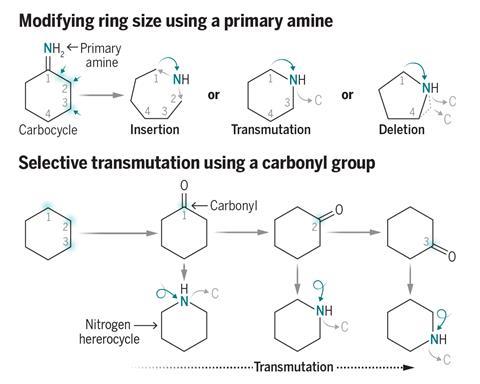
Skeletal editing techniques typically face challenges with aliphatic compounds and can only execute a single transformation, notes Zhang. He elaborates that their innovative method is capable of yielding a diverse range of nitrogen-containing heterocycles from primary amines via a common imino ether intermediary. ‘From the same starting material, we can produce [products] B, C, D, and beyond,’ states Zhang. He aspires that this will expedite drug discovery by simplifying the process of incorporating these rings into molecules and investigating how they influence the attributes of drug compounds. ‘I aim to expand the limits of skeletal editing,’ he adds.
Central to the team’s reaction is a hypervalent iodine catalyst, which possesses a central iodine atom with three substituents. Zhang mentions that this catalyst is a gentle reagent and can be synthesized with ease; the team has so far generated the iodine compound at a 100g scale.
In the reaction, the iodine catalyst initially oxidizes the amine into an imine. The ring then reorganizes by incorporating the imine nitrogen into the ring to produce a nitrilium ion – a protonated nitrile. A methoxy anion from the solvent or the iodine catalyst subsequently attacks the nitrilium to form an imino ether.
Zhang indicates that this intermediate interacts with a variety of both nucleophiles and electrophiles, such as water, azides, alkynes, benzynes, amines, and carboxylic acids. This enables the team to synthesize an array of nitrogen heterocycles of varying sizes. Depending on the reagent employed, the ring can expand, contract, or simply replace a carbon atom with a nitrogen.
‘This [work] is a splendid illustration of utilizing an abundant compound – amino-bearing cycloalkanes and n-alkanes – and integrating nitrogen,’ states Sarpong. He adds that ‘the [team] also reports an exceptionally remarkable carbon deleting amination, which enriches my thinking about what may be achievable’.
Zhang’s team is currently aiming to extend their reaction to functional groups beyond amines. He notes they are also looking for an alternative reagent with the same reactivity as their iodine-based compound, as iodine’s substantial mass indicates