**Transforming Click Chemistry: The Emergence of Copper(I)-Catalyzed Allene–Ketone Addition (CuAKA)**
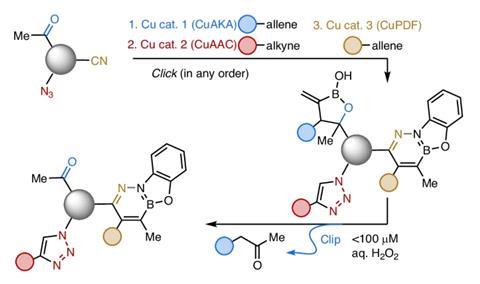
A significant advancement in the area of chemistry, the Copper(I)-Catalyzed Allene–Ketone Addition (CuAKA) reaction introduces a fresh frontier in click chemistry by generating a distinctive reversible carbon–carbon bond under biologically pertinent conditions. Historically, click chemistry has been recognized for its capacity to produce strong and permanent bonds quickly and selectively, even within complex biological settings. Nevertheless, the enduring nature of these bonds can hinder progress in domains such as drug delivery and responsive biomaterials, where reversible connections are frequently essential.
Amir Hoveyda, an esteemed chemist from the University of Strasbourg and Boston College, confronts this obstacle by demonstrating how C–C bonds, usually thought to be inappropriate for click chemistry, can be engineered to supply both durability and reversibility. This innovation contests the long-held view that the most efficient click reactions are those that generate unbreakable linkages. Rather, CuAKA enables a potent yet reversible bond, broadening the arsenal available for crafting functional molecules.
CuAKA permits the effective formation of carbon–carbon bonds via carbonyl addition in aqueous environments, a procedure once believed to be incompatible with the rapid and selective requirements of click chemistry. This reaction not only withstands the presence of complex biomolecules but also allows the amalgamation of drug-like components with peptides vital for therapeutic purposes.
The novelty resides in the reaction’s two-step mechanism, which entails the cleavage of a π-bond and the formation of a C–C bond, uniquely facilitated by its copper catalyst. It presents unparalleled opportunities for establishing new pathways in click coupling, a potential emphasized by Yimon Aye at the University of Oxford. The capability to sever the newly established link at physiological temperatures using minimal amounts of hydrogen peroxide introduces a flexible aspect to this reaction, although it necessitates thorough biological evaluation and authentication.
In a broader context, the ramifications of CuAKA are far-reaching. Within drug delivery systems, it offers a method by which pharmaceutical conjugates can sustain stability in the body until they encounter oxidative environments, such as cancerous tissues, where they can effectively dispense their payload. In the realm of chemical biology, investigators can leverage CuAKA to install and subsequently selectively eliminate probes or labels. Furthermore, the reaction’s compatibility with established copper-catalyzed click methodologies boosts its applicability in integrated molecular systems, paving the way for pioneering materials science applications, including responsive polymers and networks.
Hoveyda stresses the straightforwardness and cost-efficiency of the catalyst necessary for CuAKA, highlighting the robustness and swiftness of the reaction, which can be conducted at ambient temperatures without strict stipulations for air or moisture regulation. This breakthrough signifies a major advancement in the click chemistry field, unveiling new possibilities and indicative of the immense potential that continues to be unraveled in this arena.