Researchers in China have shown that one metallaaromatic framework can transform into another via a ring contraction reaction. Haiping Xia of Xiamen University, who led the work with Ming Luo of Southern University of Science and Technology, says this is the first aromatic framework rearrangement to be reported in an organometallic system.
The framework starts off as a metallaindenol that includes alkyne, allene, and secondary alcohol functional groups, and an osmium atom. Upon exposure to acid, the osmaindenol contracts to form an osmapentalene. The team calculated nucleus-independent chemical shift (Nics) values and performed an analysis of the anisotropy of the induced current density (Acid) for simplified models of the osmaindenol and osmapentalene; their results indicated that the osmapentalene was more aromatic than the osmaindenol and hence an increase in aromaticity drives the ring contraction reaction.
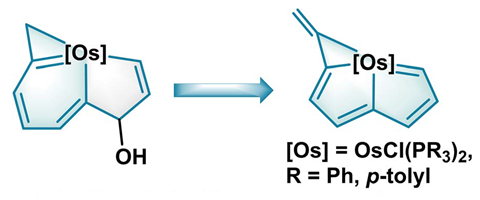
However, aromaticity continues to stir up debate. Theoretical chemist Cina Foroutan-Nejad, of the Institute of Organic Chemistry of the Polish Academy of Sciences, is not fully convinced by the researchers’ Nics and Acid data: ‘When you introduce heavy atoms to a molecule, you introduce several effects which are not related to the ring current. For example, heavy elements have a very strong local circulation of electron density induced by an external magnetic field. This local circulation often interferes with the identification of ring current.’ He would have liked the researchers to look at how relativity affected the molecules’ aromaticity. ‘When you have a heavy element like osmium, you cannot neglect relativity because it has a huge effect on chemical shifts.’
Xia, Luo and colleagues also used density functional theory (DFT) to investigate the ring contraction mechanism. The protonated osmaindenol reacts to form a mixture of two intermediates, one of which leads to an osmaindene and the other to the more stable osmapentalene. DFT also predicted that the reaction to form the osmapentalene would release hydrogen gas. ‘We employed gas chromatography to detect the presence of hydrogen gas in situ and were surprised to find that it did indeed exist,’ notes Xia. ‘This serves as an excellent example of how DFT-guided discovery can aid in understanding complex reaction processes.’
The osmium atom underpins the reshaping process. ‘In organic systems, the acid-induced indenol derivatives can generate 8π-electron antiaromatic intermediates, which further produce the corresponding SN1 substitution products. In contrast, the [osmaindenol] did not undergo a SN1 reaction with nucleophiles but transformed to the [osmapentalene],’ notes Xia. ‘The reason for this difference lies in the ability of transition metals to act as electron reservoirs, which is not possible in organic systems. This unique property of transition metals allows for novel and unexpected reactions to occur.’
‘Traditional synthetic methods for metallaaromatics are typically performed in a direct manner… This work provides an alternative approach that could inspire researchers to explore new synthetic routes for constructing aromatic metallacycles,’ concludes Xia.