A natural photoenzyme found in algae has been artificially evolved to perform an unnatural and entirely new function for the first time. The work, which converted fatty acid decarboxylase (FAP) into new-to-nature radical photocyclases that catalyse the formation of carbon rings, demonstrates how photoenzymes can be evolved and repurposed for biocatalytic applications.
FAP enzymes, which are activated by blue light, were first discovered in 2017 in green microalga and shown to produce hydrocarbons by catalysing the removal of carboxyl groups from long-chain fatty acid substrates. As one of only three known classes of natural photoenzymes, researchers have been keen to exploit FAP’s potential and produce environmentally-friendly natural products, including carbon-neutral hydrocarbons for fuels, chemicals and cosmetics.
‘In general, there is a dearth of biocatalytic carbon–carbon bond forming reactions,’ says Yang Yang at the University of California, Santa Barbara, US. ‘To expand the utility of biocatalysts, it is thus imperative to discover more enzymes that can make carbon–carbon bonds, which comprise the molecular framework of all organic compounds.’
Intrigued by FAPs’ potential, Yang and his colleagues were curious to see if they could engineer them to expand their catalytic repertoire and perform entirely different unnatural reactions for carbon–carbon bond formation. Now, Yang’s team, along with researchers at the University of Pittsburgh, US, have done just that by evolving a FAP into radical photocyclase variants using directed evolution – a high-throughput technique that mimics natural selection by subjecting a gene to successive rounds of mutagenesis until the desired function is reached.
However, it was not without challenges. ‘Directed evolution of FAPs was not easy at the beginning of our project [due to their limited stability for high-throughput techniques],’ explains Yang. ‘A talented postdoc of my group, Shuyun Ju, spent several months establishing a feasible experimental workflow. He identified a few key factors that played an important role in higher-throughput screening with photobiocatalytic functions.’
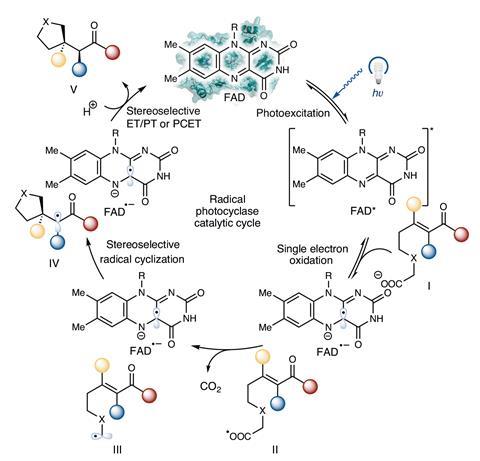
Subsequently, through rounds of directed evolution, the researchers were able to suppress the native enzyme’s decarboxylase activity while simultaneously promoting cyclisation of an emerging, and photoenzymatically formed, carbon-centred radical onto an electron-deficient alkene. The result was the production of carbon-containing rings, comprising either five or six carbons.
‘We found we can access all the four stereoisomers in radical cyclisation, with three of these stereoisomers being prepared with excellent selectivities,’ says Yang. ‘The excellent tunability of fatty acid photodecarboxylases we uncovered surprised us and demonstrated the potential of these natural photoenzymes.’
‘Remarkably, the reactions proceed in high yield and enantioselectivity with broad substrate scope,’ comments biocatalysis expert Nicholas Turner at the University of Manchester, UK. ‘This interesting study further extends the emerging class of new-to-nature biocatalysts that operate via the generation of free-radical intermediates.’
‘Obviously FAP does indeed have an enormous potential for light-assisted environmentally friendly stereoselective synthesis of small molecules and that photoenzymatic synthesis in general is the way to go,’ says Pavel Müller who co-discovered FAP at the University of Paris-Saclay, France. ‘Of the three known classes of natural photoenzymes, FAP is clearly the best and potentially most versatile chassis on which to build new constructs and try new reactions.’