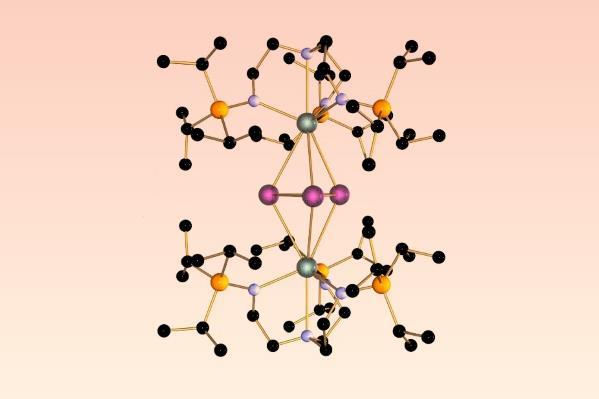
In a revolutionary advancement, researchers have created new complexes that incorporate aromatic three-atom bismuth rings stabilized by heavy actinide elements like uranium and thorium. This signifies the heaviest and one of the most distinctive instances of all-metal rings, challenging the conventional view that aromaticity is solely a characteristic of carbon-based compounds.
Traditionally, the idea of aromaticity has been intimately linked with delocalized organic molecules, epitomized by benzene. However, the field of chemistry is broadening beyond these established definitions, as demonstrated by various species that display aromatic traits without conforming to the classic framework. Remarkably, in 2024, researchers successfully created a four-membered bismuth entity, marking the birth of the first all-metal ring possessing aromatic bonding features.
This scientific pursuit has now progressed with the introduction of ‘inverted sandwich’ complexes. These complexes feature a three-membered bismuth ring, which is uniquely coordinated by two ligands containing heavy actinide elements like uranium or thorium. Consequently, these all-metal rings are the largest known analogs of aromatic three-membered rings to this point.
Comprehensive examination of the bismuth entity has uncovered a ring current similar to that which occurs in benzene. This effect is attributed to the overlapping σ-orbitals that enable the movement of electrons around the ring, thus contesting the longstanding notion that delocalized π electrons are essential for aromaticity.
Steve Liddle, from the University of Manchester, UK, who led this investigation, stressed the wider implications of this finding. He pointed out that such species serve as a powerful reminder of the universal principles of chemical bonding, which transcend the limits of carbon chemistry.