Researchers have achieved a remarkable advancement in organic chemistry by integrating a silver catalyst with a chiral sulfur reagent to stereoselectively aminate unactivated C–H bonds. This innovation facilitates late-stage modifications of various alkanes and drug-like compounds, increasing molecular complexity in a stereochemically-regulated fashion.
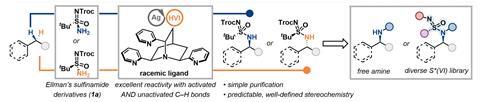
Historically, C–H amination techniques have concentrated on substrates featuring activated C–H bonds, frequently necessitating harsh conditions, excessive reagents, or costly metals such as rhodium. Nevertheless, the group led by Tuan Anh Trinh at the University of Wisconsin-Madison has introduced a groundbreaking method that employs a silver catalyst combined with a chiral sulfinamide-type reagent. This technique focuses on the less reactive, unactivated C–H bonds, resulting in the formation of a sulfonimidamide—a derivative of sulfonamide that includes an additional nitrogen group. Following this, treatment with triflic acid produces a free amine.
The method effectively aminates a range of alkane and benzylic C–H bonds, alongside intricate molecules such as steroids and diterpenes. The chirality of the sulfur(VI) reagent controls the production of specific diastereomers, allowing for precise management of the reaction’s stereochemistry. Derek Hu, another member of the team, notes silver’s distinctive abilities in creating various complexes, permitting the fine-tuning of reactivity and selectivity through ligand modification.
Their research pinpointed a quinuclidine ligand with pyridine substituents as the most effective, yielding high outputs and an equal ratio of diastereomers. Trinh highlights that this ligand’s combination of rigidity and bulkiness is essential for achieving reaction selectivity.
Peter Zhang from Boston College stresses the importance of understanding the catalytic mechanism and the characteristics of the silver-nitrogen intermediate. Gaining such knowledge will not only improve understanding of the system but will also assist in crafting the next generation of asymmetric C–H amination catalysts.
The applicability of the sulfonimidamide intermediate encompasses further functionalization with alkyne, pyridine, and cyanide groups, which hold significant value in the pharmaceutical sector due to their medicinal characteristics. Hu acknowledges the potential industrial relevance of sulfonimidamides, proposing that broadening access to these groups could profoundly affect drug development.
Zhang characterizes the research as a groundbreaking platform approach, merging late-stage functionalization, upgrading alkane feedstock, and stereochemically defined amine synthesis. He observes that this advancement illustrates the efficacy of ligand design, demonstrating how silver, although a lesser-explored metal, can become the basis for a highly proficient catalytic system with appropriate ligand engineering.