Recently, chemists have introduced a groundbreaking gas-phase nucleophilic substitution reaction in which the molecule experiences a distinctive reorientation prior to the nucleophile’s attack, thus preserving the original configuration of the molecule. Theoretical chemist Gábor Czakó, who was not part of the research, commented on the intricate nature of such reactions, implying they could be more complex than what is typically conveyed in education.
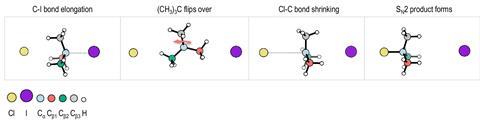
Bimolecular nucleophilic substitution, or SN2, is a fundamental reaction mechanism in chemistry that generally involves a nucleophile targeting the back face of a central carbon, leading to a configuration change known as Walden inversion. Nonetheless, other pathways such as double-inversion or front-side attack mechanisms have been noted that maintain the original configuration of the molecule.
Roland Wester’s research team, in collaboration with colleagues from the Dalian Institute of Chemical Physics, uncovered a new mechanism involving tert-butyl iodide and chloride ions. In this ‘flip-over’ mechanism, rather than the usual inversion, the molecule flips, enabling the chloride ion to strike the same face from which the iodine departs, thus preserving the configuration of the molecule. This phenomenon was observed by directing chloride ions at tert-butyl iodide at specific angles and analyzing the velocities and directions of the resulting ejected ions.
Computational studies employing a 39-dimensional potential energy surface grounded in density functional theory facilitated the comprehension of this intricate process. Through thorough experimentation, it was determined that the flip-over mechanism manifested in approximately 1% of instances, with its occurrence rising to 7% at elevated collision energies, which correspondingly lowered the rate of SN2 reactions while enhancing bimolecular eliminations.
Despite the high collision energies being unlikely in liquid environments, this discovery challenges the traditional view of SN2 reactions as merely inversion processes. The research team intends to delve deeper by examining more complex molecules and the influence of solvation on such reactions, aspiring to determine if the flip-over mechanism could be more widely applicable. Wester highlights that this discovery emphasizes the intricate nature of chemical reactions and questions longstanding beliefs.