New Studies Reveal Aluminium’s Capabilities in Catalysis
Recent developments in the field of chemistry have uncovered aluminium’s ability to replicate the reactivity of transition metals, presenting a sustainable and economical substitute for facilitating chemical reactions. Given that aluminium is among the most plentiful elements in the Earth’s crust, its application in place of less common metals could greatly enhance catalysis.
Historically, aluminium has been recognized for its +3 oxidation state; however, recent studies have brought attention to its +1 oxidation state, which has been largely ignored since the stable aluminium(I) compound was first isolated 35 years ago. New investigations have illustrated the distinct reactivity of aluminium(I).
Innovative research conducted at King’s College London, Trinity College Dublin, and the Southern University of Science and Technology showcases the reactive properties of aluminium(I). A group headed by Clare Bakewell and Tobias Krämer created aluminium atom clusters, while Xin Zhang and Liu Leo Liu formulated a redox-active aluminium species that oscillates between +1 and +3 states, facilitating alkyne reactions.
Rebecca Melen from Cardiff University commends these investigations for highlighting aluminium’s capability to replicate transition metal reactivity. “Main-group chemistry is experiencing thrilling advancements,” she remarks, emphasizing aluminium(I)’s bond activation potentials.
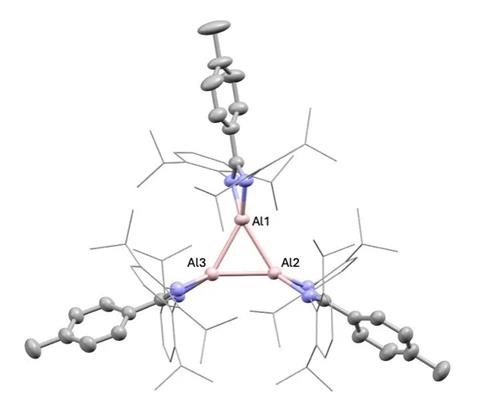
Aluminium Clusters
Bakewell’s group adeptly isolated a distinctive trimer, comprising three aluminium atoms in the +1 state, activating dihydrogen, alkynes, and benzene. Remarkably, subjecting the trimer to ethylene produced color changes, signaling reactive transformations. They identified a five-membered ring of aluminium and ethylene, a structural innovation.
Bakewell stresses that aluminium(I)’s varied reactivity in contrast to transition metals presents new avenues for chemical pathways. The team’s research enhances low-valent aluminium chemistry, paving the way for main-group redox chemistry with clusters.
A Comprehensive Catalytic Cycle
Liu and Zhang introduced an aluminium catalyst capable of a full oxidation state cycle between +1 and +3, which is vital for the Reppe reaction. This is in stark contrast to earlier aluminium catalysts, which depended on Lewis acidity and remained in the +3 state.
The research underscores the challenges and successes associated with achieving aluminium redox catalysis, which has traditionally been considered challenging for main-group elements. Modifying ligand geometry was essential for stabilizing the aluminium(I) species, facilitating continuity in the catalytic cycle.
The Future of Aluminium Chemistry
Liu and Bakewell intend to broaden the applications of their discoveries. Liu seeks to investigate single-electron processes and wider aluminium-based catalytic platforms. Bakewell examines the influence of ligand variations on trimer reactivity.
The field is progressing swiftly, with breakthroughs such as the isolation of a dialumene radical cation illustrating aluminium’s redox adaptability and exceptional reactivity. As Bakewell observes, “The chemistry of low oxidation state aluminium is abundant,” with new findings arising consistently.