Researchers in China have made a significant advance by developing the initial polymer networks crosslinked through pnictogen bonds. The process of crosslinking polymer chains is a frequently utilized technique to improve the mechanical and functional characteristics of materials. Permanent covalent bonds ensure stability, while reversible bonds offer systems the capability to reorganize under different circumstances. This flexibility is especially beneficial for materials intended for self-healing or responsiveness to external stimuli.
A variety of supramolecular interactions are utilized to construct crosslinked polymers, with hydrogen-bonded networks being the most prevalent. However, hydrogen bonds frequently exhibit limited strength and compatibility with solvents. Chalcogen and halogen bonding provide promising alternatives in the field of dynamic polymer materials, though achieving the right balance of properties remains a challenge.
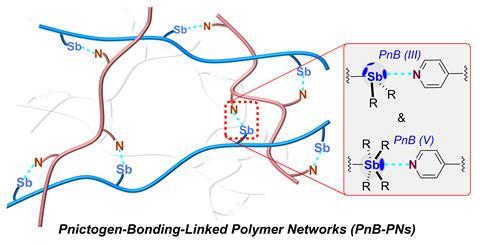
Pnictogen bonding was recently defined as interactions that involve an electrophilic region on a pnictogen atom and a nucleophilic site in a molecular entity. Under the leadership of Wei Wang, a research team from Zhengzhou University in China took advantage of this type of bonding to create materials capable of self-healing, even in underwater environments.
These groundbreaking materials employ interactions between antimony and pyridine-functionalized polymer chains, with antimony chosen for its strong Lewis acidity in comparison to other pnictogens. By altering the oxidation state of the antimony, researchers were able to fine-tune the system, given that the bond strengths of pnictogens differ between Sb(iii) and Sb(v) centers.
Pnictogen bond donors typically feature more accessible σ-holes than other non-covalent donors, enhancing bonding directionality and fostering more complex structural arrangements within the polymer, which allows for improved control over its properties. Additionally, the water resistance of pnictogen bonds significantly broadens the potential applications of these polymers.