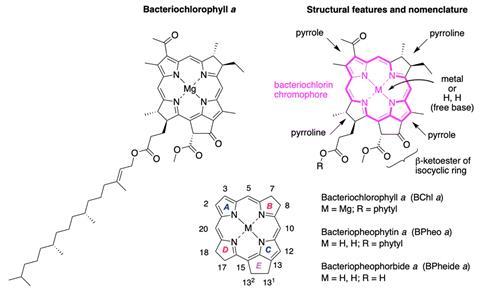
After almost ten years of intense work, a group of chemists in the United States, led by Jonathan Lindsey from North Carolina State University, has achieved the groundbreaking total synthesis of bacteriochlorophyll a. This intricate molecule, essential in photosynthesis, has been challenging for chemists to synthesize due to its complex structure, which includes four stereocentres in its chromophore and an epimerisable β-ketoester within an isocyclic ring. These characteristics make it susceptible to breaking down into simpler derivatives, creating significant synthetic obstacles.
In the past, efforts to synthesize this compound have been limited. A prominent attempt in 1960 underscored the challenges tied to the synthesis of chlorophyll a, establishing a standard that very few have approached since. Yet, Lindsey’s team, in the face of challenges including global chemical shortages due to the Covid-19 pandemic and the heartbreaking loss of team member Khiêm Châu Nguyễn, remained determined. They committed to overcoming experimental challenges and confirming the stereochemistry of the synthesized structures, which took more time than expected.
The synthesis entailed a multi-step procedure, initiated by asymmetric Michael addition to set the stereochemistry of pyrroline ring precursors. This was succeeded by the combination of molecular halves through Knoevenagel condensation and a ring closure using Nazarov cyclisation and electrophilic aromatic substitution. This technique not only constructs the complicated macrocycle effectively but also maintains the desired stereochemical arrangement.
Respected colleagues within the scientific community, like Ana Moore from Arizona State University, have praised this accomplishment as a “synthetic tour de force,” acknowledging the possibility for this efficient method to be modified for the synthesis of additional photosynthetic pigments. Looking ahead, Lindsey plans to enhance this synthesis, making it more efficient and versatile, thereby expanding the potential for a deeper exploration of photosynthetic mechanisms.